Services to industry
- Home /
- Services to industry

Services to Industry
We support industry segments that include life sciences, food, biotechnology, medical devices and pharmaceutical companies, writing agencies, scientific journals and publishers. Our business model allows the medical and scientific writers to be integral part of research industries, writing agencies, journals and publishers.
Though we primarily focus on writing scientific or medical content, our multi-talented writers are equipped with skills such as adequate project and stakeholder management, knowledge on health regulatory changes, working in a corporate environment, business acumen, awareness of electronic data management systems, flexibility to quickly adapt to the client requirements, etc.
Our services for life sciences, food, biotechnology, medical devices and pharmaceutical companies include writing and submitting both medical communications and regulatory submission documents.
While, our services for writing agencies, scientific journals and publishers include copy editing, peer review and peer review management, journal specific formatting and managing outsourced writing and editing assignments.
Services for life sciences, food, biotechnology, medical devices and pharmaceutical companies
We combine proficient understanding of local and global regulatory and ethical issues related to medical writing with efficiency and cost-effectiveness of our global network of medical writers. Our medical writer’s talents and efforts are coordinated to meet your deadlines and quality through efficient SOPs and project management tools. We help our customers to bring out a wide range of regulatory, clinical, scientific and marketing communication documents as per their requirement allowing them to focus on their core competency areas. You can rely on our customer-centric, robust processes and turnaround time to bring your product faster into the market.
We gratify all your regulatory writing and medical communication needs. All our regulatory documents are fully compliant with the respective ICH and other regulatory guidelines. We prepare high-quality scientific publications based on all available ethical guidelines and journal/conference instructions.
Medical Communication Documents / Services
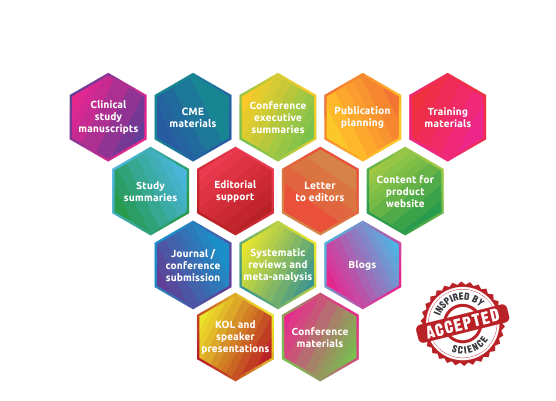
Regulatory Submision Documents

Services for writing agencies, conference organizers, scientific journals and publishers

Are you a scientific writing services provider? Do you need support in completing your writing projects on time to serve your clients better?
We can provide you a wide array of cost effective offerings such as project management, copy editing, QC checks, journal-specific formatting, and manuscript technical requirements check.
Are you a journal publisher and finding difficulty in conducting peer review, formatting and proof reading?
SciWrite Global can provide you exclusive journal management system to enable you to publish issues on time with better quality and visibility. We also assist societies, organizations and individuals in setting up and establishing new journals, obtaining impact factor, improving the geographical reach and visibility, and indexing in reputed scientific databases.
We support the conference organizers in effectively managing their scientific activities. We support them in soliciting scientific abstracts, their peer review, designing the abstract book and its publication.
We also write and edit textbooks, Continued Medical Education (CME) programs, patient education materials, and e-learning modules. In addition, we have extensive experience in providing high-quality copyediting services for STM journals and publishers. We partner with the publishing industry to provide services such as journal-specific formatting, professional language editing, manuscript technical requirements check, peer review process management, etc. We also offer custom services.
We provide high-quality, cost-effective and successful scientific writing solutions to academia and industry. Please contact managingeditor@sciwriteglobal.com for more information on the way we work, our customized prices and new offers.
